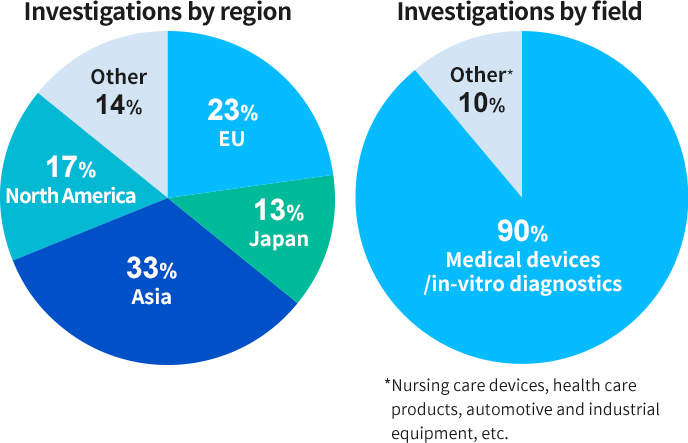
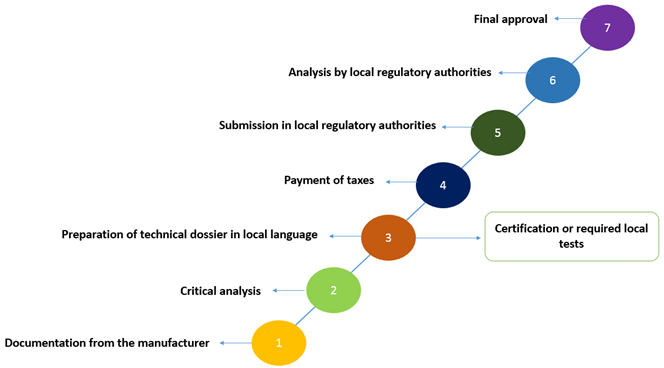
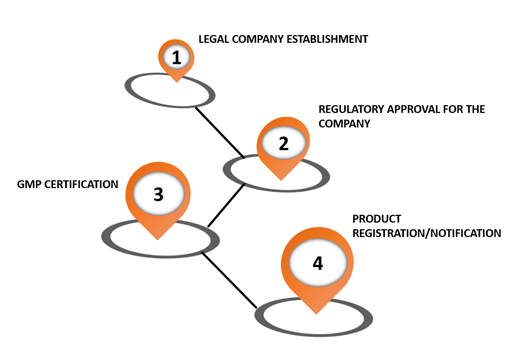
Q&A REGULATORY AFFAIRS IN LATIN AMERICA. DEFINITION OF MEDICAL DEVICE Product, equipment, device, material, article or system of Medical, Odontological. - ppt download

Latin America: Understanding Regulatory Compliance Requirements Across the Life Science Industry by ComplianceOnline - Issuu

PPT – Latin America APEC Funded Medical Device Regulatory Seminar PowerPoint presentation | free to view - id: 4afea-ZDc1Z

Colombia, Mexico and Cuba promote the creation of the Regulatory Agency for Drugs and Medical Devices of Latin America and the Caribbean | Ministerio de Relaciones Exteriores de Cuba

Latin America Offers Welcoming Entry Market Amid Tighter EU Regulations — bioaccess.™: CRO. REGULATORY. COMMERCIALIZATION